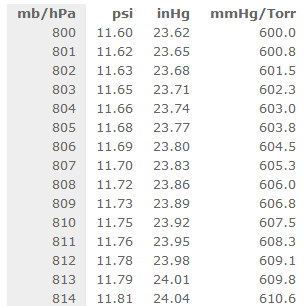
Take a sample of gas at STP 1 atm and 273 K and double the temperature.ĭoubling the temperature, likewise doubled the pressure. This law holds true because temperature is a measure of the average kinetic energy of a substance when the kinetic energy of a gas increases, its particles collide with the container walls more rapidly and exert more pressure. 1 mm of mercury is the pressure exerted by a 1 inch high column of mercury, now defined as 133.322387415m pascals. Where #P_1# stands for the initial pressure of the gas, #T_1# stands for the initial temperature, #P_2# stands for the final pressure of the gas, and #T_2# stands for the final temperature. The law has a simple mathematical form if the temperature is measured on an absolute scale, such as in kelvins. Pressure and temperature will both increase or decrease simultaneously as long as the volume is held constant. Simplified, this means that if you increase the temperature of a gas, the pressure rises proportionally. 1 Torr is defined exactly equal to 1 760 of one standard atmosphere. In other words, Gay-Lussac's Law states that the pressure of a fixed amount of gas at fixed volume is directly proportional to its temperature in kelvins. Table 4: Difference between mmHg and Torr mmHg: Torr: 1 mmHg is defined as the pressure exerted by 1 mm of a column of mercury. Gay-Lussac’s Law is an ideal gas law where at constant volume, the pressure of an ideal gas is directly proportional to its absolute temperature. To convert bar to atm, multiply the bar value by 0.98692326671 or divide by 1.01325. Millimeters of mercury to Millibar formula. 1 mm of mercury is the pressure exerted by a 1 inch high column of mercury, now defined as 133.322387415m pascals. #V_2 = ? color(white)(mml)n_2 = "0.500 mol + 0.250 mol = 0.750 mol"# More information: Millimeters of mercury. If we add 0.250 mol of gas at the same pressure and temperature, what is the final total volume of the gas?
Thus, its molar volume at STP is 22.71 LĪ 6.00 L sample at 25.0 ☌ and 2.00 atm contains 0.500 mol of gas. One mole of an ideal gas occupies 22.71 L at STP.
#V/n = k#, where #k# is a proportionality constant.Įqual volumes of hydrogen, oxygen, or carbon dioxide contain the same number of molecules. #V ∝ n#, where #V# is the volume, and #n# is the number of moles. When the tank cools to 20.0 ☌, the pressure of the gas is 30.0 atm. The gas is placed into a tank under high pressure. It does not depend on the sizes or the masses of the molecules. 500.0 liters of a gas are prepared at 700.0 mmHg and 200.0 ☌. The volume increases as the number of moles increases.
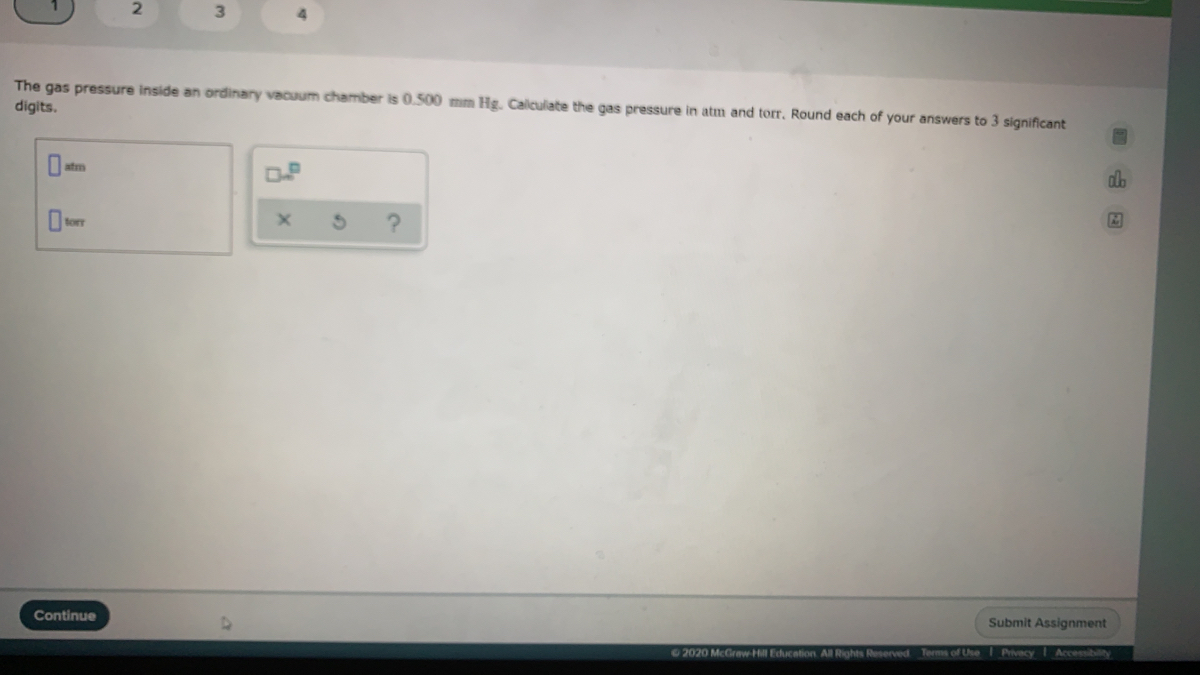
Another statement is, "Volume is directly proportional to the number of moles." Air pressure is expressed using a variety of measures including atm (atmospheres) Torr (1 atm 760 Torr) pounds force per square inch (1 atm 14.6959 psi) mm Hg (1 atm.
